Why Bacteriostatic Water is Essential for Peptide Reconstitution
Mar 17th 2026
In peptide research, reconstitution is the critical first step that turns stable lyophilized powder into a usable solution. The solvent you choose directly impacts sterility, peptide stability, and the reliability of your results over multiple draws. Bacteriostatic water has become the standard in most research settings because it maintains sterility through repeated vial access, preserves peptide integrity without interference, and supports efficient multi-dose protocols. Using anything less reliable often leads to contamination, reduced potency, or inconsistent data, issues that can compromise entire experiments. Here’s a clear breakdown of why bacteriostatic water is preferred, how it compares to alternatives, and the precise steps to reconstitute peptides correctly and safely.
What is Peptide Reconstitution?
Peptide reconstitution dissolves the freeze-dried (lyophilized) powder back into a liquid form suitable for precise dosing in experimental models. The powder form protects the delicate amino acid chains during transport and storage by removing water and preventing oxygen and moisture from entering. To use it, you add a calculated volume of solvent that must dissolve the peptide uniformly without denaturing the peptide or introducing contaminants.
The goal is a clear, stable solution at a known concentration (e.g., 1 mg/mL or 2 mg/mL) that allows accurate pipetting or syringe draws. Poor technique, like vigorous shaking, using the wrong solvent, or ignoring sterility, can cause foaming (which risks oxidation), precipitation (insoluble aggregates), or rapid degradation from bacterial byproducts. When done correctly, reconstituted peptides maintain potency for weeks under proper storage, enabling long-term studies without constant re-preparation.
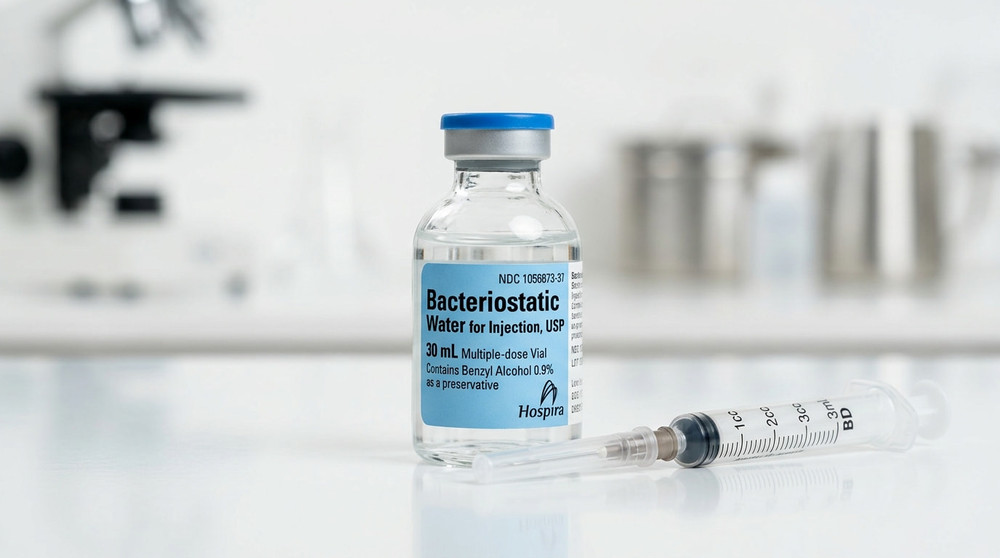
What is Bacteriostatic Water?
Bacteriostatic water for injection, USP, is sterile water containing 0.9% benzyl alcohol as a preservative. The benzyl alcohol inhibits bacterial reproduction (bacteriostatic, not bactericidal), preventing contamination in multi-dose vials without aggressively disrupting most biological molecules. Produced to pharmaceutical standards (e.g., by Hospira, a Pfizer subsidiary), it comes in 30 mL multiple-dose vials with semi-rigid polyolefin construction designed for repeated needle punctures while maintaining seal integrity. It’s non-pyrogenic, clear, and pH-neutral enough (~5.7–7.0) for broad compatibility.
Why Bacteriostatic Water is Preferred for Peptides
1. Maintains Sterility
Each vial puncture introduces a small contamination risk from the needle or the environment. Without a preservative, bacteria can proliferate quickly even in trace amounts, leading to cloudy solutions or enzymatic breakdown of the peptide. The 0.9% benzyl alcohol creates an environment hostile to microbial growth, allowing safe repeated access for 28 days post-opening when refrigerated properly.
2. Protects Peptide Stability
Peptides are sensitive to pH shifts, oxidation, and bacterial enzymes. Contaminated solutions accelerate degradation; benzyl alcohol prevents that cascade. At this low concentration, it doesn’t alter peptide folding or bioactivity for the vast majority of research compounds unlike harsher preservatives or mismatched solvents that might cause precipitation or reduced efficacy.
3. Multi-Use Convenience
Protocols often require daily or frequent draws over weeks. Single-use vials force wasteful re-opening of new solvent bottles each time. Bacteriostatic water’s design supports up to 28 days of reliable use from one vial, reducing cost, plastic waste, and preparation time while keeping concentrations consistent across experiments.
4. Safe & Compatible with Most Peptides
It works cleanly with popular research peptides like BPC-157, TB-500, CJC-1295 (with or without DAC), Ipamorelin, IGF-1 LR3, GHK-Cu, and more. Dissolution is typically complete without clumping, and the benzyl alcohol rarely triggers adverse reactions at standard concentrations. For rare ultra-sensitive cases, alternatives like acetic acid exist, but bacteriostatic water covers 90%+ of needs without complications.
Bacteriostatic Water vs Other Solvents
Sterile Water
Sterile water is preservative-free and intended strictly for single-use. Once opened, any contamination can lead to bacterial growth within hours to days. For peptides needing multiple draws, this is a major risk, as reconstituted solutions can degrade or become unusable fast. It’s fine for immediate, one-shot dilutions but unsuitable for ongoing research protocols.
Saline Solutions
0.9% sodium chloride mimics physiological conditions and is used in some clinical settings, but for peptides it introduces variables: salt can shift ionic balance, promote aggregation in hydrophobic sequences, or affect stability during storage. Bacteriostatic water avoids these extras, keeping the focus on the peptide itself.
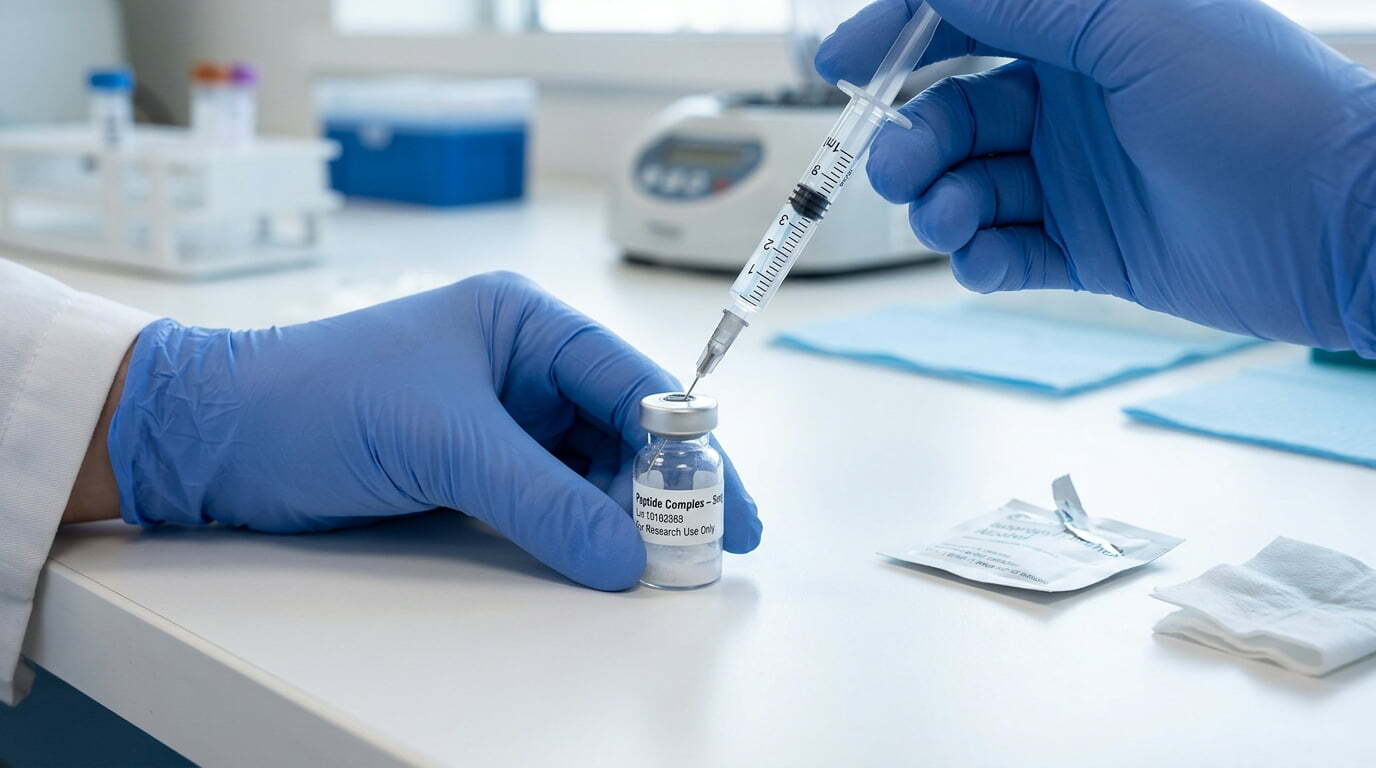
How to Reconstitute Peptides (Step-by-Step Guide)
- Prepare aseptic workspace: Wash hands, wear gloves, disinfect surfaces. Allow peptide and bacteriostatic water vials to reach room temperature (~30 minutes) to avoid thermal shock.
- Sanitize vials: Remove caps, swab rubber stoppers with 70% isopropyl alcohol, and let air-dry completely.
- Calculate volume: Target concentration matters, e.g., for a 5 mg vial, 2 mL bacteriostatic water yields 2.5 mg/mL (easy dosing math). Use peptide calculators if unsure.
- Draw solvent: Use a new sterile syringe. Pull the exact volume from the bacteriostatic water vial.
- Inject gently: Insert the needle into the peptide vial, angle to let the solvent run down the inner wall slowly, avoid direct powder blasts that cause foaming or denaturation.
- Dissolve: Swirl gently in circular motions (never shake vigorously). Most peptides dissolve in 1–5 minutes; warm slightly in hand if needed, but don’t heat externally.
- Inspect and store: Ensure a clear solution, label the vial with date, concentration, and peptide name. Refrigerate at 2–8°C immediately.
Best Practices for Using Bacteriostatic Water
- Always use fresh needles/syringes per draw, reuse increases contamination odds.
- Store unopened vials at controlled room temperature (20–25°C); refrigerate only after opening or mixing peptides (cold can sometimes reduce benzyl alcohol effectiveness over very long periods).
- Discard opened bacteriostatic water after 28 days; appearance alone isn’t enough; preservative efficacy wanes.
- Maintain strict aseptic technique: No finger contact with stoppers, no cross-contamination between vials.
- For peptides prone to oxidation, minimize air exposure during mixing and consider nitrogen purging if your lab setup allows.
- Track batches: Record solvent lot numbers alongside peptide details for traceability.
Why Choose Hospira Bacteriostatic Water (Arpovo Health)
Hospira remains the benchmark for pharmaceutical-grade bacteriostatic water, sterile, non-pyrogenic, with reliable 0.9% benzyl alcohol that labs and clinics trust worldwide. Arpovo Health supplies authentic 30 mL Hospira vials at competitive pricing ($16.99), in stock with fast USA shipping (free on all orders, bulk discounts for 2+). Their dedicated Hospira Bacteriostatic Water page details everything, while the broader solutions page and diluent category cover matching sterile options if needed. Sealed packaging and Pfizer-backed quality make it a no-brainer for researchers prioritizing consistency.
Trusted Peptide Sources for Research
Pairing quality solvent with quality peptides is non-negotiable. Patriot Peptides offers a focused lineup of high-purity, lyophilized research compounds, BPC-157 (5 mg), CJC-1295 variants, Ipamorelin, IGF-1 LR3, GHK-Cu (up to 50 mg), and others, all produced under strict standards with emphasis on lab-only use. Browse the full, well-organized selection at the Patriot Peptides shop. Their research guidelines page is a solid resource too: it covers proper storage (temperature/light/moisture control), labeling for traceability, structural notes (e.g., DAC vs non-DAC), and quality verification, all reinforcing best practices that align perfectly with careful reconstitution.
Conclusion
Bacteriostatic water is more than a convenience; it’s a safeguard that protects peptide integrity, prevents contamination pitfalls, and enables efficient, multi-dose research without constant re-prep. Choosing poorly here undermines everything downstream. Opt for proven pharmaceutical-grade products like Hospira from Arpovo Health, follow rigorous techniques, and source peptides from reliable vendors, and your data becomes more reliable, your protocols more repeatable, and your time far better spent on actual science.
FAQ
What is bacteriostatic water used for?
Primarily for reconstituting lyophilized peptides and other research compounds requiring multiple sterile draws over time.
Can bacteriostatic water be reused?
Yes, safely for up to 28 days after opening due to the benzyl alcohol preservative.
How long does bacteriostatic water last after opening?
28 days when refrigerated and handled aseptically; discard after regardless of appearance.
Is bacteriostatic water safe for peptides?
Yes, standard and compatible for most research peptides, with negligible interference at 0.9% benzyl alcohol.
What is the difference between sterile and bacteriostatic water?
Sterile water lacks preservatives and is single-use only (high contamination risk if reused). Bacteriostatic water includes 0.9% benzyl alcohol to inhibit bacterial growth, supporting multi-dose use for weeks.